Əhaliyə xəbərdarlıq: Bu gün 15:00-dan sonra...

İranda bir ayda 80 məhbus edam edildi

Bu imtahanların tarixi açıqlandı

Tramp bu halda yenidən müharibəyə başlayacaq

Amerikalı jurnalist Putindən vətəndaşlıq istədi

Final oyunu: Azərbaycan Ukraynaya qarşı

Putin müntəzəm olaraq xokkey oynayır - Peskov

Dubay şahzadəsinin azərbaycanlı arvadı saxlanıldı

Qadın sürücü qəza törətdi: azyaşlı övladı öldü

1562-ci gün: Rusiyanın itkiləri yeniləndi

İranın İraqdakı qruplaşması silahı yerə qoydu

Azərbaycan nefti bahalaşdı

Əlilliyi olan şəxslərlə bağlı bu qaydalar təsdiqləndi

ABŞ iştahını itirəndə İrəvanla buna son qoyacaq - Rusiya

Rusiya bir gecədə 272 PUA vurdu

Sovetin əfsanəsi qayıtdı: Yeni “Volqa” fantastik qiymətə... - Foto

Qəzalı körpü söküldü: Yenisi inşa edilir - Video

Bitkoinin qiyməti 62 min dollardan aşağı düşdü

Livan və İsrail atəşkəs barədə razılığa gəldilər

Rusiya "Geran-4"ü işə sala bilər: Saatda 500 km sürət…

Rusiyadan Ukrayna ilə bağlı üç ssenari: yaxşı, pis və...
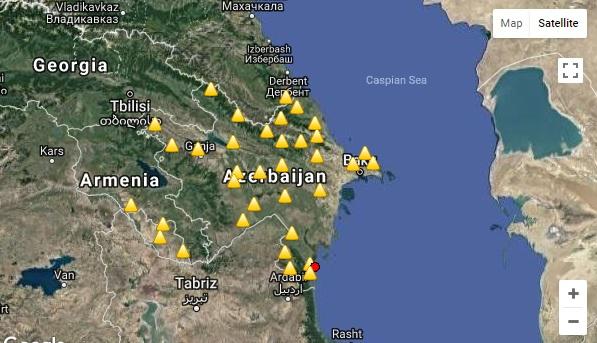
Xəzər dənizində zəlzələ baş verdi

Bu, Rusiyaya ağır başa gələcək - Zelenski

Üç ölkənin Rusiya ilə bağlı gizli planı...

ABŞ-dan İrana xəbərdarlıq: Bir qismini ləğv edəcəyik!

Kələmin əsas təhlükəsi açıqlandı

Lavrovla dəfələrlə buna görə danışmışam - Rubio

"Ford" 420 min avtomobili geri çağırır